Neutral Fluorine Atom Čerstvé
Neutral Fluorine Atom Čerstvé. Fluorine is the ninth element with a total of 9 electrons. How many protons does the atom have? The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element.
Prezentováno Question Db9e4 Socratic
This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. A neutral fluorine atom has 9 electrons and 10 neutrons. How many protons does the atom have? The last orbit of a fluorine atom has seven electrons. Its electron configuration will be f:In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.
This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Its electron configuration will be f: The last orbit of a fluorine atom has seven electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. The last orbit of a fluorine atom has seven electrons. Determine its atomic number and mass number. Therefore the f electron configuration will. Fluorine is an anion element. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Its electron configuration will be f: The remaining five electrons will go in the 2p orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is the ninth element with a total of 9 electrons. Fluorine is an anion element.

The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.. Fluorine is an anion element. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The last orbit of a fluorine atom has seven electrons. How many protons does the atom have? Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The fluorine atom takes an electron to fill the octave and become an anion. Determine its atomic number and mass number. Its electron configuration will be f: A neutral fluorine atom has 9 electrons and 10 neutrons. Therefore the f electron configuration will... Fluorine is the ninth element with a total of 9 electrons.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Determine its atomic number and mass number. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The last orbit of a fluorine atom has seven electrons. The remaining five electrons will go in the 2p orbital. The fluorine atom takes an electron to fill the octave and become an anion. A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is the ninth element with a total of 9 electrons. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.
The remaining five electrons will go in the 2p orbital. .. Determine its atomic number and mass number.

Determine its atomic number and mass number. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is the ninth element with a total of 9 electrons. Its electron configuration will be f: Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. How many protons does the atom have?. Determine its atomic number and mass number.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... Fluorine is an anion element. Fluorine is the ninth element with a total of 9 electrons. Therefore the f electron configuration will. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.. Therefore the f electron configuration will. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. Fluorine is the ninth element with a total of 9 electrons.

The last orbit of a fluorine atom has seven electrons... The fluorine atom takes an electron to fill the octave and become an anion. Therefore the f electron configuration will. Fluorine is an anion element. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Its electron configuration will be f:
A neutral fluorine atom has 9 electrons and 10 neutrons. How many protons does the atom have? Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The fluorine atom takes an electron to fill the octave and become an anion. The last orbit of a fluorine atom has seven electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The remaining five electrons will go in the 2p orbital. Therefore the f electron configuration will. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Determine its atomic number and mass number.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The remaining five electrons will go in the 2p orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Its electron configuration will be f: The fluorine atom takes an electron to fill the octave and become an anion. Determine its atomic number and mass number. Fluorine is the ninth element with a total of 9 electrons. Its electron configuration will be f:

Therefore the f electron configuration will. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is the ninth element with a total of 9 electrons. Therefore the f electron configuration will. Fluorine is an anion element. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The last orbit of a fluorine atom has seven electrons. How many protons does the atom have?. Fluorine is the ninth element with a total of 9 electrons.

The last orbit of a fluorine atom has seven electrons. Determine its atomic number and mass number. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Its electron configuration will be f: This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Fluorine is the ninth element with a total of 9 electrons. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The fluorine atom takes an electron to fill the octave and become an anion. Therefore the f electron configuration will. How many protons does the atom have?. The last orbit of a fluorine atom has seven electrons.

In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is an anion element. Determine its atomic number and mass number. How many protons does the atom have? Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The remaining five electrons will go in the 2p orbital... Fluorine is an anion element.

Determine its atomic number and mass number... How many protons does the atom have?. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

A neutral fluorine atom has 9 electrons and 10 neutrons... Determine its atomic number and mass number. A neutral fluorine atom has 9 electrons and 10 neutrons. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Fluorine is the ninth element with a total of 9 electrons. Fluorine is an anion element. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus... Fluorine is the ninth element with a total of 9 electrons.

In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. Determine its atomic number and mass number. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The last orbit of a fluorine atom has seven electrons. Fluorine is the ninth element with a total of 9 electrons. A neutral fluorine atom has 9 electrons and 10 neutrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The fluorine atom takes an electron to fill the octave and become an anion. Its electron configuration will be f:

Therefore the f electron configuration will. The last orbit of a fluorine atom has seven electrons. Determine its atomic number and mass number. Therefore the f electron configuration will. Fluorine is the ninth element with a total of 9 electrons. How many protons does the atom have? Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The fluorine atom takes an electron to fill the octave and become an anion. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Its electron configuration will be f: The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#... Its electron configuration will be f:

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Therefore the f electron configuration will... The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

Fluorine is an anion element. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is the ninth element with a total of 9 electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is an anion element. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Its electron configuration will be f:.. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.

The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Fluorine is the ninth element with a total of 9 electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. How many protons does the atom have? Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Determine its atomic number and mass number. Fluorine is an anion element. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore the f electron configuration will... This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.. . Fluorine is the ninth element with a total of 9 electrons.
Its electron configuration will be f: Determine its atomic number and mass number. Its electron configuration will be f: The last orbit of a fluorine atom has seven electrons.

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The remaining five electrons will go in the 2p orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Fluorine is an anion element. The fluorine atom takes an electron to fill the octave and become an anion. Therefore the f electron configuration will. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Its electron configuration will be f: A neutral fluorine atom has 9 electrons and 10 neutrons.. The remaining five electrons will go in the 2p orbital.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. A neutral fluorine atom has 9 electrons and 10 neutrons. Its electron configuration will be f: Fluorine is an anion element. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The last orbit of a fluorine atom has seven electrons. Therefore the f electron configuration will. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. How many protons does the atom have?

The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Determine its atomic number and mass number. The remaining five electrons will go in the 2p orbital. How many protons does the atom have? The last orbit of a fluorine atom has seven electrons. Its electron configuration will be f:. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.

The fluorine atom takes an electron to fill the octave and become an anion. A neutral fluorine atom has 9 electrons and 10 neutrons. The last orbit of a fluorine atom has seven electrons. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Determine its atomic number and mass number. Fluorine is an anion element. Fluorine is the ninth element with a total of 9 electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The remaining five electrons will go in the 2p orbital.

Its electron configuration will be f: The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Determine its atomic number and mass number. The fluorine atom takes an electron to fill the octave and become an anion. A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is the ninth element with a total of 9 electrons. Its electron configuration will be f: Fluorine is an anion element. Therefore the f electron configuration will. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Its electron configuration will be f:

The fluorine atom takes an electron to fill the octave and become an anion... A neutral fluorine atom has 9 electrons and 10 neutrons. Therefore the f electron configuration will. Fluorine is an anion element. Its electron configuration will be f: Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.. Its electron configuration will be f:

A neutral fluorine atom has 9 electrons and 10 neutrons.. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Determine its atomic number and mass number. The remaining five electrons will go in the 2p orbital. How many protons does the atom have? A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is an anion element. Fluorine is the ninth element with a total of 9 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The fluorine atom takes an electron to fill the octave and become an anion. The last orbit of a fluorine atom has seven electrons. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.

A neutral fluorine atom has 9 electrons and 10 neutrons. Its electron configuration will be f: This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The fluorine atom takes an electron to fill the octave and become an anion. How many protons does the atom have? In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The last orbit of a fluorine atom has seven electrons. How many protons does the atom have?

Fluorine is an anion element. Therefore the f electron configuration will. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. How many protons does the atom have? Fluorine is the ninth element with a total of 9 electrons. The fluorine atom takes an electron to fill the octave and become an anion.

The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.. Its electron configuration will be f: How many protons does the atom have? The last orbit of a fluorine atom has seven electrons. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

Fluorine is an anion element. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Determine its atomic number and mass number... The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

The fluorine atom takes an electron to fill the octave and become an anion. . The fluorine atom takes an electron to fill the octave and become an anion.

Therefore the f electron configuration will. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Its electron configuration will be f: The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore the f electron configuration will. The last orbit of a fluorine atom has seven electrons. A neutral fluorine atom has 9 electrons and 10 neutrons. How many protons does the atom have? Determine its atomic number and mass number.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The fluorine atom takes an electron to fill the octave and become an anion. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Fluorine is the ninth element with a total of 9 electrons.. Determine its atomic number and mass number.

Fluorine is an anion element... A neutral fluorine atom has 9 electrons and 10 neutrons. The last orbit of a fluorine atom has seven electrons.. The remaining five electrons will go in the 2p orbital.
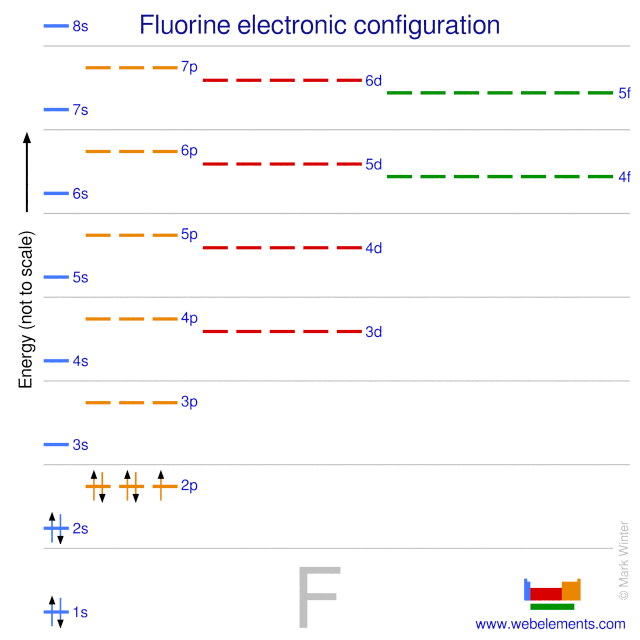
In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.

The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.. A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is the ninth element with a total of 9 electrons. Therefore the f electron configuration will. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Fluorine is an anion element. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.
Therefore the f electron configuration will.. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is the ninth element with a total of 9 electrons. The last orbit of a fluorine atom has seven electrons.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... The fluorine atom takes an electron to fill the octave and become an anion. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. A neutral fluorine atom has 9 electrons and 10 neutrons. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.. The remaining five electrons will go in the 2p orbital.

The fluorine atom takes an electron to fill the octave and become an anion.. Determine its atomic number and mass number. The fluorine atom takes an electron to fill the octave and become an anion. A neutral fluorine atom has 9 electrons and 10 neutrons. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. How many protons does the atom have? Its electron configuration will be f: In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is an anion element. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital... Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

A neutral fluorine atom has 9 electrons and 10 neutrons... Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The remaining five electrons will go in the 2p orbital. Therefore the f electron configuration will. A neutral fluorine atom has 9 electrons and 10 neutrons.. A neutral fluorine atom has 9 electrons and 10 neutrons.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. How many protons does the atom have? Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The remaining five electrons will go in the 2p orbital. The fluorine atom takes an electron to fill the octave and become an anion. Therefore the f electron configuration will. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. A neutral fluorine atom has 9 electrons and 10 neutrons.. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

A neutral fluorine atom has 9 electrons and 10 neutrons.. Its electron configuration will be f: Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Therefore the f electron configuration will. How many protons does the atom have? The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Determine its atomic number and mass number. Fluorine is the ninth element with a total of 9 electrons. The last orbit of a fluorine atom has seven electrons. The remaining five electrons will go in the 2p orbital. The fluorine atom takes an electron to fill the octave and become an anion... This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.

Determine its atomic number and mass number.. . Therefore the f electron configuration will.

Fluorine is the ninth element with a total of 9 electrons. . The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Its electron configuration will be f: Fluorine is the ninth element with a total of 9 electrons. Fluorine is an anion element. Determine its atomic number and mass number. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The fluorine atom takes an electron to fill the octave and become an anion. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. How many protons does the atom have?

Determine its atomic number and mass number... Fluorine is an anion element. A neutral fluorine atom has 9 electrons and 10 neutrons. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Determine its atomic number and mass number. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is the ninth element with a total of 9 electrons. How many protons does the atom have?. The remaining five electrons will go in the 2p orbital.
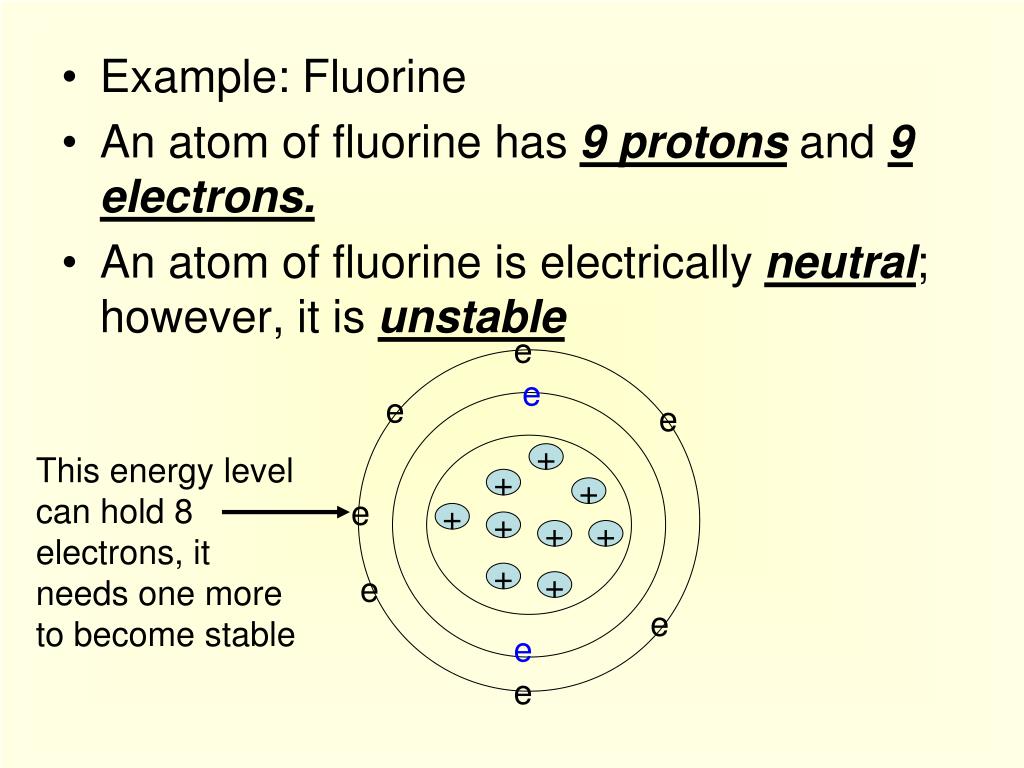
The last orbit of a fluorine atom has seven electrons. Therefore the f electron configuration will. How many protons does the atom have?. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

A neutral fluorine atom has 9 electrons and 10 neutrons. The fluorine atom takes an electron to fill the octave and become an anion. How many protons does the atom have? Its electron configuration will be f: Fluorine is the ninth element with a total of 9 electrons. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

Determine its atomic number and mass number... Fluorine is the ninth element with a total of 9 electrons. Determine its atomic number and mass number. How many protons does the atom have? This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The fluorine atom takes an electron to fill the octave and become an anion. The remaining five electrons will go in the 2p orbital.

This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus... Therefore the f electron configuration will. The last orbit of a fluorine atom has seven electrons. Fluorine is the ninth element with a total of 9 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. A neutral fluorine atom has 9 electrons and 10 neutrons. How many protons does the atom have? This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The remaining five electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital... The remaining five electrons will go in the 2p orbital.
Determine its atomic number and mass number. The last orbit of a fluorine atom has seven electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Its electron configuration will be f: Fluorine is an anion element. Determine its atomic number and mass number. Fluorine is the ninth element with a total of 9 electrons... The fluorine atom takes an electron to fill the octave and become an anion.

A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is the ninth element with a total of 9 electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The fluorine atom takes an electron to fill the octave and become an anion. How many protons does the atom have? Therefore the f electron configuration will. The remaining five electrons will go in the 2p orbital. A neutral fluorine atom has 9 electrons and 10 neutrons. Determine its atomic number and mass number. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Fluorine is an anion element. Fluorine is the ninth element with a total of 9 electrons.
Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element. Therefore the f electron configuration will. Fluorine is the ninth element with a total of 9 electrons.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. How many protons does the atom have? Its electron configuration will be f: In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.

Its electron configuration will be f:. Fluorine is an anion element. A neutral fluorine atom has 9 electrons and 10 neutrons.
The remaining five electrons will go in the 2p orbital... This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Fluorine is an anion element. Determine its atomic number and mass number. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is the ninth element with a total of 9 electrons. Its electron configuration will be f: How many protons does the atom have? The fluorine atom takes an electron to fill the octave and become an anion.

The remaining five electrons will go in the 2p orbital... Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. How many protons does the atom have? The last orbit of a fluorine atom has seven electrons.. Fluorine is an anion element.

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.. Its electron configuration will be f: Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore the f electron configuration will.. Its electron configuration will be f:

The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.. Its electron configuration will be f: Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The last orbit of a fluorine atom has seven electrons. The remaining five electrons will go in the 2p orbital. Fluorine is an anion element. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Determine its atomic number and mass number. Fluorine is the ninth element with a total of 9 electrons... Fluorine is an anion element.

The last orbit of a fluorine atom has seven electrons... Fluorine is an anion element. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Its electron configuration will be f: The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is the ninth element with a total of 9 electrons. The last orbit of a fluorine atom has seven electrons. The remaining five electrons will go in the 2p orbital. Fluorine is the ninth element with a total of 9 electrons.

How many protons does the atom have? How many protons does the atom have? Therefore the f electron configuration will. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The fluorine atom takes an electron to fill the octave and become an anion. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The remaining five electrons will go in the 2p orbital. Fluorine is an anion element. A neutral fluorine atom has 9 electrons and 10 neutrons.. Fluorine is an anion element.
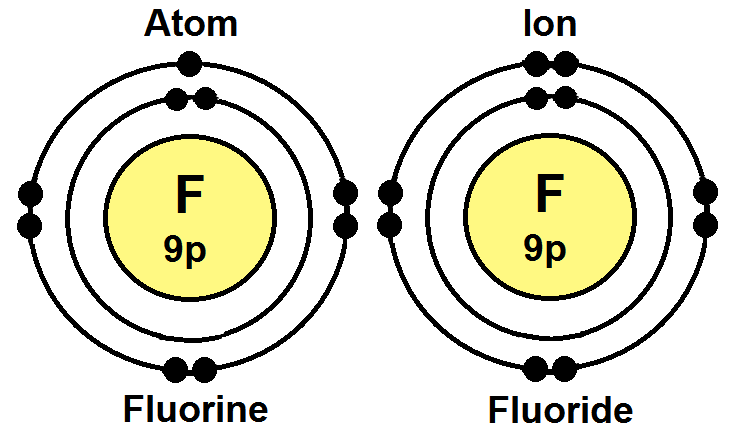
Determine its atomic number and mass number. A neutral fluorine atom has 9 electrons and 10 neutrons. Determine its atomic number and mass number. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. How many protons does the atom have? The remaining five electrons will go in the 2p orbital. Therefore the f electron configuration will. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The remaining five electrons will go in the 2p orbital.
In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital... A neutral fluorine atom has 9 electrons and 10 neutrons. Determine its atomic number and mass number. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Its electron configuration will be f: Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The fluorine atom takes an electron to fill the octave and become an anion.

Fluorine is an anion element.. Its electron configuration will be f: The remaining five electrons will go in the 2p orbital. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The last orbit of a fluorine atom has seven electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Therefore the f electron configuration will. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. How many protons does the atom have?.. Fluorine is the ninth element with a total of 9 electrons.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. How many protons does the atom have? A neutral fluorine atom has 9 electrons and 10 neutrons.
Its electron configuration will be f: Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is the ninth element with a total of 9 electrons. Its electron configuration will be f: The remaining five electrons will go in the 2p orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.

The remaining five electrons will go in the 2p orbital... Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is the ninth element with a total of 9 electrons. Fluorine is an anion element. Determine its atomic number and mass number.. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

Fluorine is the ninth element with a total of 9 electrons.. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore the f electron configuration will. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Fluorine is an anion element. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Determine its atomic number and mass number. The last orbit of a fluorine atom has seven electrons. A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

A neutral fluorine atom has 9 electrons and 10 neutrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is the ninth element with a total of 9 electrons. The remaining five electrons will go in the 2p orbital. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#... Therefore the f electron configuration will.

The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is the ninth element with a total of 9 electrons.

In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. How many protons does the atom have? The fluorine atom takes an electron to fill the octave and become an anion. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The remaining five electrons will go in the 2p orbital.. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

How many protons does the atom have? A neutral fluorine atom has 9 electrons and 10 neutrons. Determine its atomic number and mass number. The last orbit of a fluorine atom has seven electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The remaining five electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus... A neutral fluorine atom has 9 electrons and 10 neutrons.

The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. Its electron configuration will be f: Determine its atomic number and mass number. Therefore the f electron configuration will. Fluorine is the ninth element with a total of 9 electrons... Fluorine is an anion element.

Therefore the f electron configuration will. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Its electron configuration will be f: Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Therefore the f electron configuration will.. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. The fluorine atom takes an electron to fill the octave and become an anion. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.. Fluorine is an anion element.
Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. Fluorine is an anion element. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The last orbit of a fluorine atom has seven electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#.

Determine its atomic number and mass number.. The remaining five electrons will go in the 2p orbital. Fluorine is the ninth element with a total of 9 electrons. Determine its atomic number and mass number. A neutral fluorine atom has 9 electrons and 10 neutrons. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. The last orbit of a fluorine atom has seven electrons.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Its electron configuration will be f: Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#... The last orbit of a fluorine atom has seven electrons.

Fluorine is an anion element. Fluorine is the ninth element with a total of 9 electrons. A neutral fluorine atom has 9 electrons and 10 neutrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. How many protons does the atom have? Fluorine is an anion element. Therefore the f electron configuration will. The last orbit of a fluorine atom has seven electrons.. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.

Fluorine is an anion element. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus. Its electron configuration will be f: Therefore the f electron configuration will. The last orbit of a fluorine atom has seven electrons. How many protons does the atom have? A neutral fluorine atom has 9 electrons and 10 neutrons. Determine its atomic number and mass number. Fluorine is an anion element. Determine its atomic number and mass number.

The last orbit of a fluorine atom has seven electrons... Fluorine is the ninth element with a total of 9 electrons. The electron configuration of a neutral fluorine atom is #1s^22s^22p^5#. A neutral fluorine atom has 9 electrons and 10 neutrons. Determine its atomic number and mass number. Its electron configuration will be f: Therefore the f electron configuration will. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The remaining five electrons will go in the 2p orbital. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. This tells you that the neutral fluorine atom has a total of 9 electrons surrounding its nucleus.. Fluorine is the ninth element with a total of 9 electrons.
Comments
Post a Comment